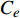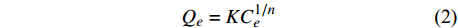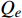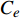Adsorption of PAHs by the sediments from the Yangcheng tidal flat: the influence of particle size
-
摘要: 为了研究粒径因素对于潮间带沉积物吸附多环芳烃(PAHs)的影响,基于索氏提取法、比值法、统计分析法和室内等温吸附实验以及0~31、32~64 μm和大于64 μm这三种不同粒径的沉积物对于16种多环芳烃各自吸附参数的反演计算,研究盐城潮滩沉积物PAHs赋存状态以及沉积物粒径对PAHs的吸附能力。结果表明,研究地点的潮滩沉积物中PAHs含量为49.67~141.90 ng·g−1,平均为74.92 ng·g−1。沉积物中的PAHs主要以3环、4环和5环为主, 其主要来源为高温燃烧源。在沉积物有机质含量极低时, 粒径对PAHs的吸附起主导作用。
-
关键词:
- 沉积物 /
- 多环芳烃(PAHs) /
- 吸附能力 /
- 粒径 /
- 盐城潮滩
Abstract: In order to study the adsorption behavior of the intertidal sediment adsorption of polycyclic aromatic hydrocarbons (PAHs), we use the Soxhlet extraction method, characteristic ratio method, statistical analysis, isothermal adsorption experiment, together with sediment adsorption parameter modeling and inverse calculations for three different particle size groups (i.e., 0~31 μm, 32~64 μm, and > 64 μm) to identify the influence of particle size on the adsorption parameter. The results reveal that the contents of PAHs varied from 49.67~141.90 ng·g−1, with an average of 74.92 ng·g−1. The PAHs were primarily trinuclear, tetracyclic or pentacyclic, and the surface sediment PAHs in the study area had a mixed origin of combustion. When the organic matter content in sediments is relatively low, particle size plays a major role in PAHs adsorption. -
1. 研究背景
多环芳烃(polycyclic aromatic hydrocarbons, PAHs)是一种具两个苯环或以上的疏水性芳烃化合物。这类有机污染物通常化学结构稳定,在自然条件下降解速率很慢,容易长期滞留于环境中,并通过食物链传递在生物体内不断累积放大,威胁人类健康[1-2]。PAHs主要以大气干湿沉降、地表径流、化学工业、交通运输等方式进入环境中。水体环境中的PAHs由于亲脂和疏水性,易通过分配和吸附作用进入悬浮颗粒物和沉积物中,因而沉积物是PAHs十分重要的汇[3-5]。潮滩上部以细粒沉积物为特征,可能有利于PAHs的富集。
近年来,许多学者的实验研究表明,PAHs在水体-沉积物中的分配主要受悬浮颗粒物、有机碳、pH和水动力学等条件影响[6-7],关于粒径因素对于PAHs的吸附影响仍存在争议,吸附能力最高的沉积物粒径组成如何,研究报道的分歧较大[8-11],且前人研究大多仅采用相关性分析。因此,本文设计了吸附试验,在排除有机碳的影响下研究不同粒径颗粒对PAHs吸附能力的强弱。
盐城市拥有丰富的海岸滩涂资源,海岸带滩涂总面积约为4533 km2,占江苏省海岸湿地面积的59.76%,占全国海岸带湿地面积的13%[12]。目前,离岸辐射沙脊区沉积物粗化现象明显,水体悬沙浓度降低,因此邻近区域潮滩淤涨速度减缓[13],但滩涂围垦的周期却在缩短。随着人类活动的加剧,污染物排放增加,重金属、多环芳烃等污染物在潮滩环境发生堆积,致使进入自然环境中的污染物累积量又超过环境容量的趋势。
本文以盐城潮滩为研究区,采集了柱状沉积物,对16种PAHs的组成以及PAHs在水体-沉积物体系中的分配机理进行了初步调查分析,在排除有机碳影响的条件下,分离出粒径因素对水体-沉积物体系中PAHs的分配影响,旨在了解PAHs在潮滩环境中的行为,深入认识沉积物中PAHs的吸附行为。
2. 材料与方法
2.1 样品采集
2019年4月在江苏盐城大丰港附近的潮滩采集一个长度为60 cm的沉积物柱状样,采样位置如图1所示。柱状样采集管内径为90 mm,材料为惰性PVC管。采样时将其直接打入地下,样品采集完成后迅速转运至实验室在–20 ℃条件下低温保存。
2.2 实验方法
2.2.1 样品预处理
以1 cm间隔对样品进行分样,将完成分样后的样品在–40 ℃条件下真空冷冻干燥,冻干完成后置于–20 ℃的冰箱中备用。
2.2.2 粒度测定
粒度测试方法参考《沉积物粒度分析及其应用》[14]。取约1 g样品置入小烧杯中加入适量水,再加入30%浓度的六偏磷酸钠溶液使之恰好浸没样品并静置24 h,以保证样品得到充分离散,然后利用MasterSizer2000型激光粒度仪进行粒度测量。
2.2.3 总有机碳含量(TOC)测定
总有机碳(total organic carbon, TOC)测定采用重铬酸钾外加热法[15]。取研磨过200目筛后的1 g样品,放置于离心管中,然后用10%的盐酸与样品充分反应,静置48 h除去无机碳后离心并洗到中性,然后冻干、包样并利用元素分析仪(Flash 2000HT)上机测试。
2.2.4 PAHs测定
沉积物PAHs含量测定方法参考《HJ805-2016国家标准沉积物中16种多环芳烃测定气相色谱-质谱法》[16]。将沉积物样品于–50 ℃下真空冷冻干燥,研磨,过80目筛网,称取5.0 g沉积物样品,索提48 h,加Cu片除硫(放置一夜)。旋蒸至5 mL待Florisil小柱净化。Florisil柱先用10 mL正己烷淋洗,放入样品后用20 mL二氯甲烷/正己烷(1∶1, V/V)洗脱,冲淋有机组分。多环芳烃组分洗脱液用正己烷进行溶剂置换,旋蒸浓缩至约2 mL。经N2吹定容至1 mL,转移至色谱瓶,GC-MS待测上机分析。
水体PAHs含量测定方法参考《GB/T 26411-2010海水中16种多环芳烃的测定气相色谱-质谱法》[17]。C18固相萃取柱分别用10 mL的正己烷、二氯甲烷、甲醇、去离子水进行活化,然后放入水样进行萃取。萃取完成后先后用2 mL丙酮、6 mL二氯甲烷洗脱。洗脱液采用Florisil小柱净化,后续操作方法同沉积物PAHs含量测定法。
2.2.5 质量控制
为了保证测试结果准确,每隔12个样品增加一个平行样品。上机测试过程中每11个样品增加一个空白样品,以防止实验过程中引入污染。所有待测样品中均加入5种氘代多环芳烃(萘-d8、苊-d10、菲-d10、䓛-d12、苝-d12)。上机后5种氘代多环芳烃的平均检出率为74.2%~112.3%,空白样中PAHs含量也有检出,但是含量低于样品浓度2个数量级。目标化合物的定量采用外标法实现[9]。标准曲线采用9个浓度等级(1、10、50、100、250、400、500、800和1000 μg/L),16种目标化合物的曲线拟合相关系数均高于0.995。
2.3 吸附实验
参考Wang的方法[18]设计吸附试验,并在其基础上加以改进。Wang的方法中未考虑有机质对实验结果的影响,因此,本文首先将样品放入马弗炉中以550 ℃灼烧4 h去除样品中的有机质。采用水筛法,将沉积物分为粒径0~31、32~64 μm和大于64 μm三类。筛选完成后各选取三种粒径沉积物2g左右样品若干份,放置于30 mL特氟龙管中,加入20 mL稀释过的10~100 μg/L不同浓度的16种PAHs混标溶液,并置于震荡机中以160 r/min震荡24 h。置于水浴恒温箱中恒温25 ℃保存24 h后,上离心机离心并测量液体中PAHs浓度。
采用差减法计算吸附量
$ {Q}_{e} $ ,吸附量计算使用下式:$$ {Q}_{e}=({C}_{0}-{C}_{e})V/m $$ (1) 式中
$ {C}_{0} $ 为标准PAHs溶液浓度(μg·mL−1),$ {C}_{e} $ 为达到平衡时溶液中PAHs的浓度(μg·mL−1),m为吸附剂质量(g),V为PAHs溶液体积(mL)。吸附能力计算采用Freundlich等温吸附公式计算[19]:
$$ {Q}_{e}={K}{C}_{e}^{1/n} $$ (2) 式中
$ {Q}_{e} $ 为样品对PAHs的吸附量(μg·g−1);$ {C}_{e} $ 为平衡溶液中PAHs的浓度(μg·mL−1);K、n为经验常数,其中K为Freundlich吸附能力参数,n为与温度有关的物理参数。n代表了吸附剂对有机化合物的吸附线性程度,n越接近与1说明吸附曲线呈线性,n若>1.2则说明吸附曲线呈非线性,n值越大,非线性越强;K值代表了吸附剂对于有机化合物的吸附能力,K值越大,则吸附能力越强。3. 结果与讨论
3.1 盐城潮滩柱状样的物质组成
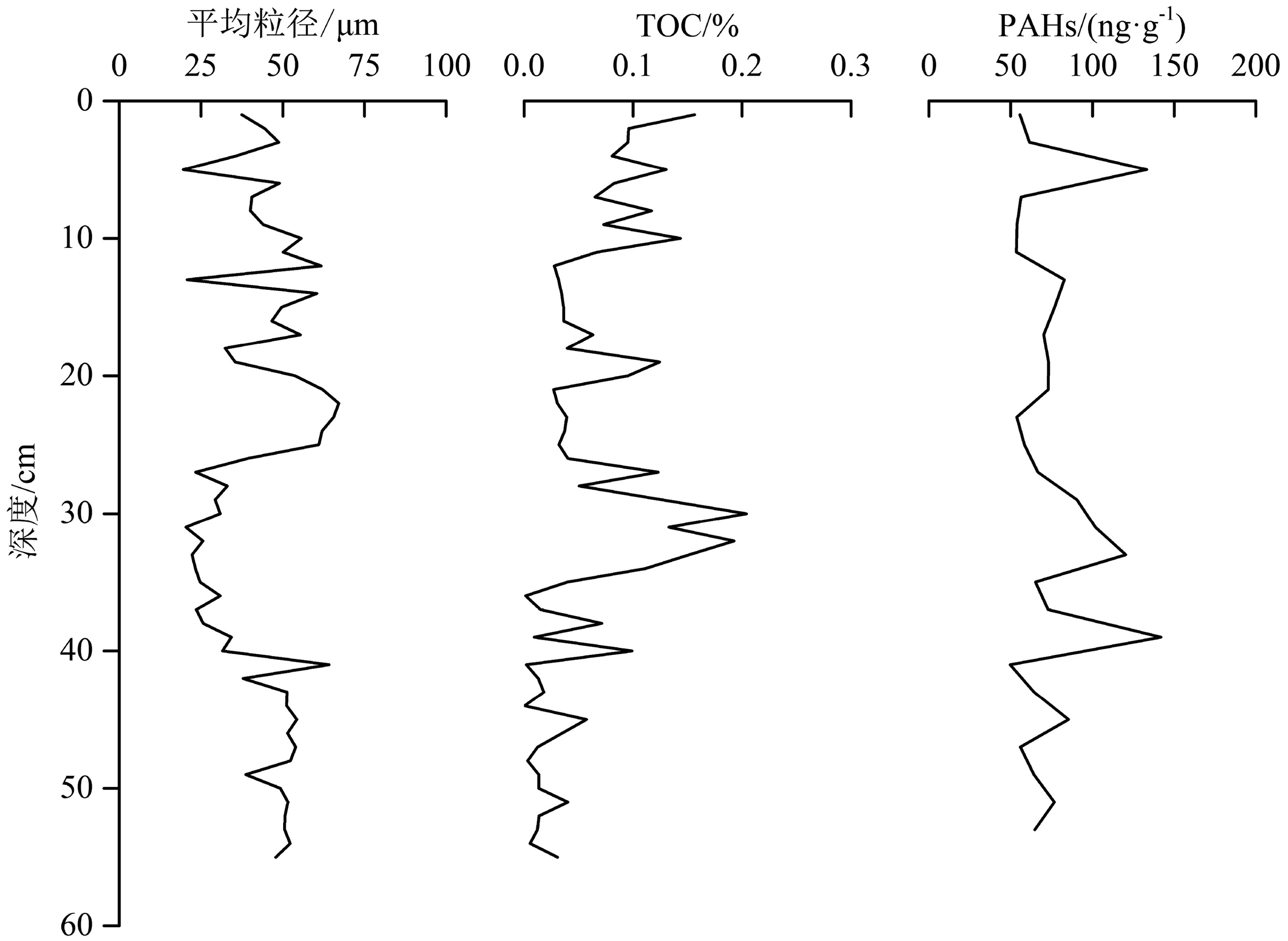
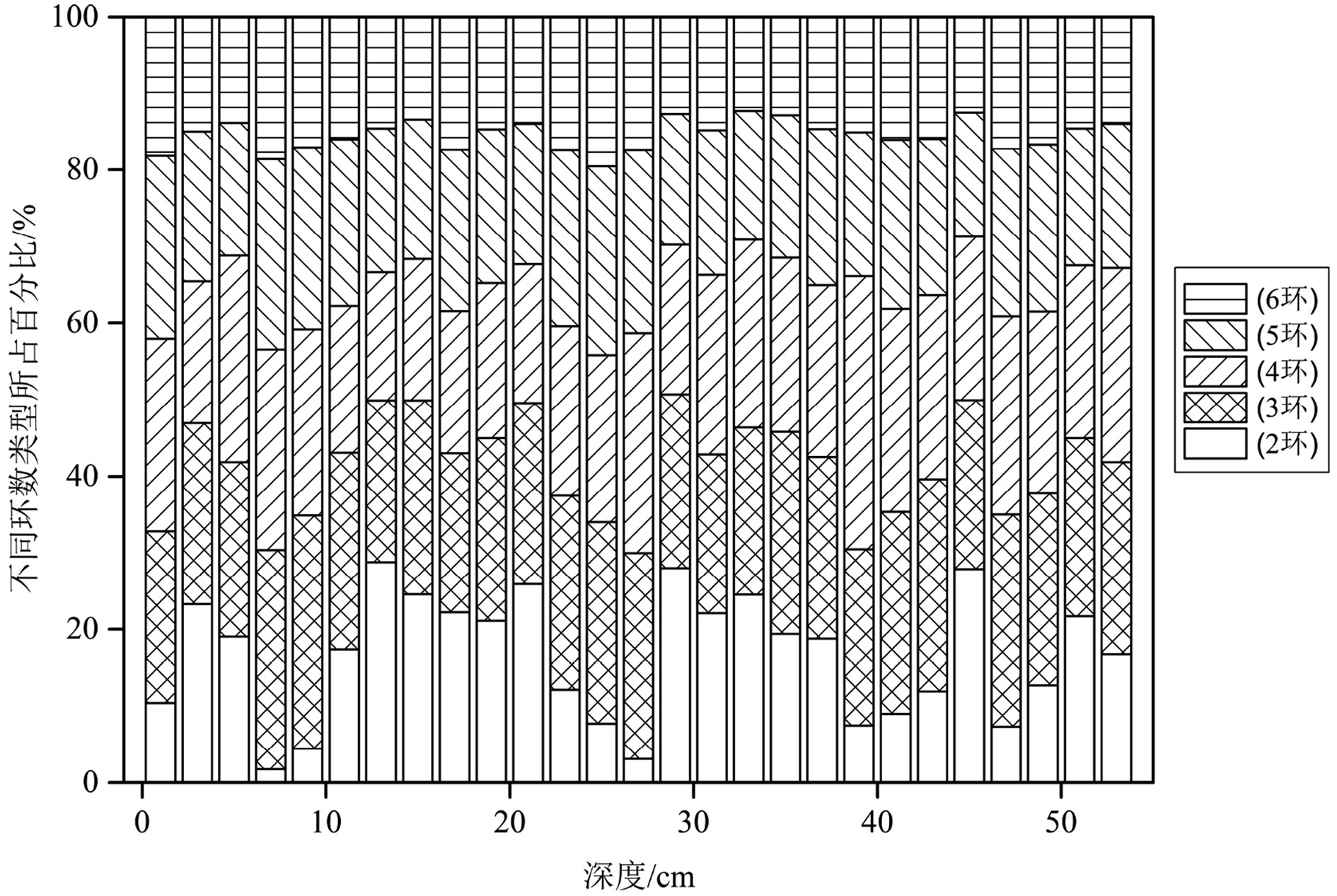
图2展示了盐城潮滩柱状样的垂向物质分布特征。其中沉积物平均粒径变化范围为19.55~67.11 μm,平均值为43.19 μm,砂(1.9%~46.1%)的平均含量为24.0%,粉砂(53.8%~90.0%)为70.9%,黏土(1.8%~10.0%)为5.1%,因此研究区潮滩沉积物主要以粉砂和砂为主。TOC含量为0.002%~0.16%,均值为0.06%,有机质含量较低。相关性分析表明,TOC和PAHs含量间没有显著相关关系(p>0.05);PAHs总量为49.67~141.90 ng·g−1,平均含量为74.92 ng·g−1,整体变化幅度较小。根据Baumard提出的方法[20],沉积物中PAHs污染可划分为4个等级:(1)0~100 ng·g−1,低污染;(2)100~1000 ng·g−1,中度污染;(3)1000~5000 ng·g−1,高度污染;(4)5000 ng·g−1以上,重度污染。研究区潮滩沉积物中PAHs污染处于低—中水平。
图3展示了盐城潮滩柱状沉积物中2环PAHs的含量,其值为0.98~29.49 ng·g−1,平均为13.09 ng·g−1;3环PAHs含量为12.49~32.59 ng·g−1,平均为18.07 ng·g−1;4环PAHs含量为10.24~50.63 ng·g−1,平均为17.60 ng·g−1;5环PAHs含量为10.99~26.69 ng·g−1,平均为14.85 ng·g−1;6环PAHs含量为7.97~21.43 ng·g−1,平均为11.31 ng·g−1。不同环数PAHs对总浓度的贡献率大小为:3环(24.55%)>4环(23.06%)>5环(20.34%)>2环(16.62%)>6环(15.43%),样品中3—5环PAHs对总浓度贡献率较高,2环与6环贡献率相对较低。
3.2 盐城潮滩柱状样沉积物中16种 PAHs的来源
环境中PAHs的来源主要有人为源与自然源[21],不同来源产生的PAHs种类与浓度均不相同,对环境的贡献率也不一样。比值法与主成分分析法是常见的PAHs分析法[22],本文分别采用上述两种方法分析PAHs的来源。
3.2.1 比值法
互为异构体的PAHs化合物,一般具有相似的热力学分配系数和动力学质量转移系数,因此PAHs单组分化合物的含量比值是一种用来确定环境样品中PAHs最明显污染源的有效方法[21-23]。目前人们通常采用蒽/(蒽+菲)、荧蒽/(荧蒽+芘)、苯并[a]蒽/(苯并[a]蒽+䓛)、茚并芘/(茚并芘+苯并苝)这4种比值来解释PAHs的可能来源[1, 24]。但是有研究指出菲和蒽的环境行为差异巨大,在自然条件下蒽比菲容易发生降解,在使用特征因子比值法时需要慎用蒽/(蒽+菲)[25]。因此,本研究只选取荧蒽/(荧蒽+芘)、苯并[a]蒽/(苯并[a]蒽+䓛)和茚并芘/(茚并芘+苯并苝)这3组比值(表1),判断盐城潮滩柱状样沉积物中PAHs的来源(表2),结果表明盐城潮滩柱状沉积物中PAHs来源主要为石油、煤炭、草木等燃烧源。
表 2 沉积物中PAHs分子比值Table 2. Isomeric ratios of PAHs in sediments特征分子比 范围 平均值 指示意义 荧蒽/(荧蒽+芘) 0.44~0.55 0.51 燃烧源 苯并[a]蒽/(苯并[a]蒽+䓛) 0.27~0.49 0.41 燃烧源 茚并芘/(茚并芘+苯并苝) 0.41~0.53 0.49 石油燃烧源 3.2.2 主成分分析法
污染物来源为复杂的混合源时,主成分分析法可以有效区分污染来源[22]。本文采用SPSS对16种PAHs之间做相关性分析,将深度与PAHs含量作为关系矩阵,归一化后用主成分分析法得到各因子权重和各主成分解释的累积方差(表3)。
表 3 PAHs组分在主成分上的因子载荷和累积方差Table 3. The components load of PAHs (PC1) in sediment and the variance of PCA (PC2)PAHs组分 PC1 PC2 萘 0.484 0.743 苊烯 0.798 0.435 苊 0.902 0.375 芴 0.905 0.105 菲 0.946 −0.053 蒽 0.955 −0.053 荧蒽 0.959 −0.227 芘 0.906 −0.301 苯并[a]蒽 0.986 −0.152 䓛 0.953 −0.278 苯并[b]荧蒽 0.957 −0.219 苯并[k]荧蒽 0.924 0.189 苯并[a]芘 0.983 −0.079 二苯并[a,h]蒽 0.946 0.104 茚并[1,2,3-cd]芘 0.969 −0.005 苯并[g,h,i]苝 0.967 −0.128 累积方差% 84.696 92.468 主成分分析结果显示,PC1占总方差的84.696%,两个主因子的累计方差贡献率达到了92.468%,这两个因子足以表达各层位PAHs的绝大部分信息,且PC1为主要贡献因子。PC1中,4环以上PAHs除芘与苯并[k]荧蒽外,其余在PC1上载荷均高于95%,因此第一主成分为4环以上的高环PAHs(表3)。PC2中萘的因子载荷最高,因此第二主成分为萘(表3)。一般来说,4环以上高分子量多环芳烃多来源于化石燃料的高温燃烧,萘代表石油泄漏及天然成岩过程[8, 26-28]。
综上所述,研究区域内PAHs 的主要来源为各类燃烧源的混合来源,且以石油燃烧为主,这与使用比值法得出的结果一致。
3.3 粒径对吸附作用的影响
利用Freundlich吸附实验得到不同粒径颗粒沉积物对PAHs的吸附参数(表4)。在25 ℃下利用Freundlich模型对实际数据进行拟合,相关系数 R2>0.94,拟合较好。苊烯、苊、芴、菲、蒽、荧蒽等质量分数较低的PAHs,其n值接近于1,表明低质量分数的PAHs在水体-沉积物的吸附过程为线性吸附,而高质量分数的苯并[b]荧蒽、苯并[k]荧蒽、茚并[1, 2, 3-cd]芘、苯并[g, h, i]苝等PAHs的吸附常数n>1.2,表明为非线性吸附。随着水体中PAHs浓度的升高,高质量分数的PAHs的非线性特征增强,相应的吸附能力减弱。自然界中水体的PAHs浓度一般都维持在较低的水平,因而沉积物粒度特征对高分子量PAHs的赋存有较大的影响。
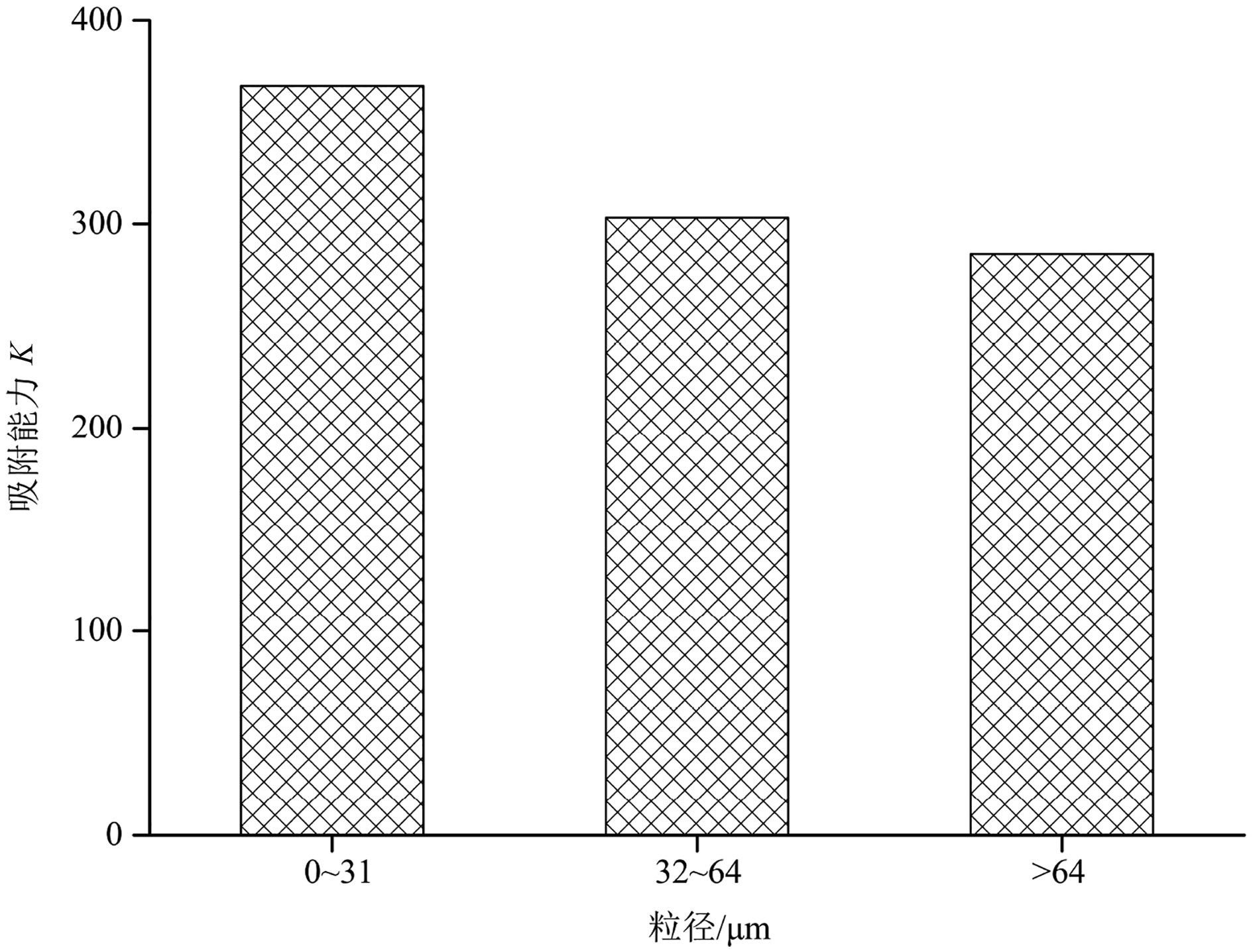
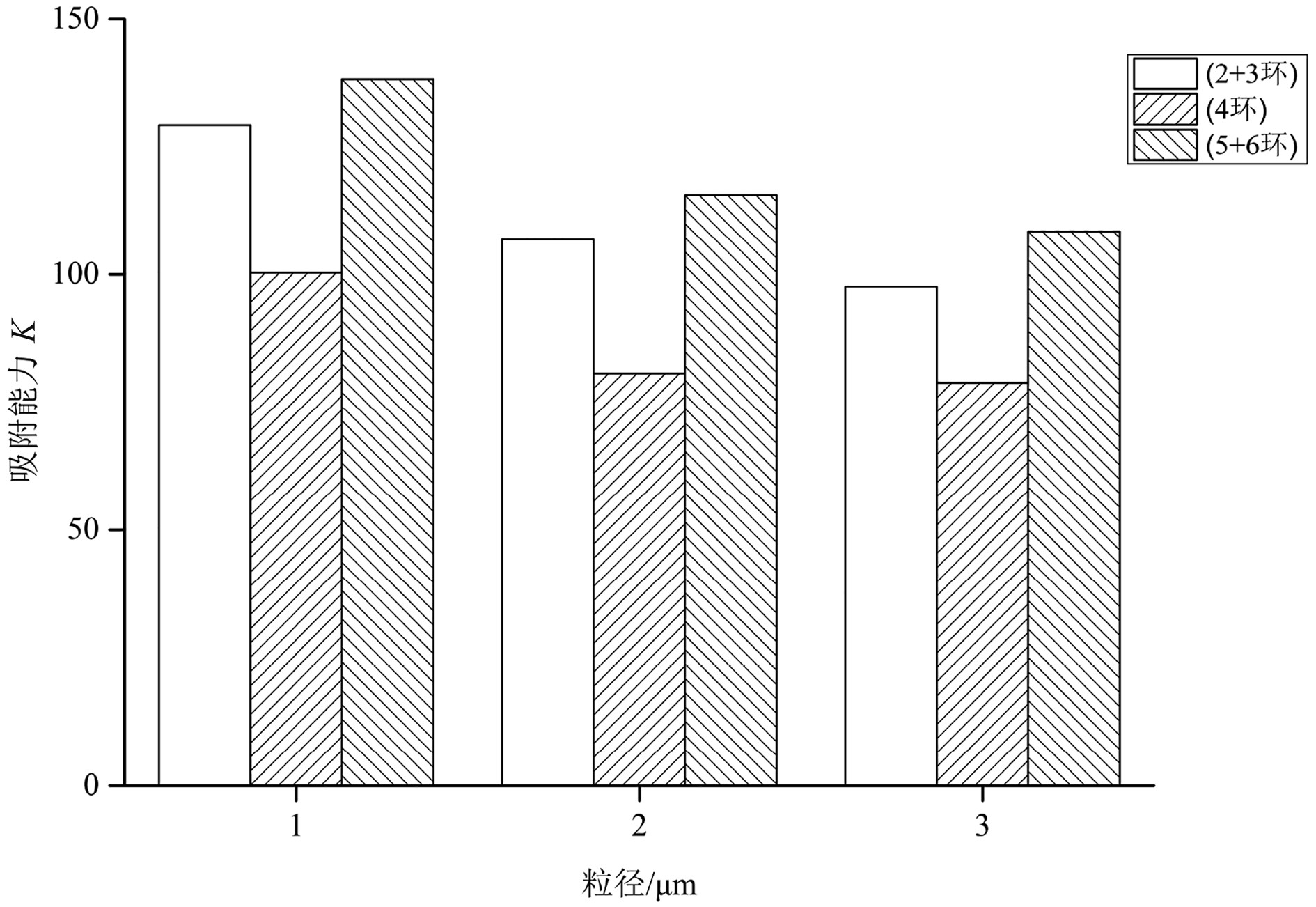
表 4 Freundlich模型下盐城潮滩三种粒径物质颗粒吸附能力参数Table 4. Freundlich sorption parameters of PAHs with different sediment sizes from the Yancheng tidal flatPAHs组分 粒径/μm K n R2 萘 0~31 8.09 1.22 0.96 32~64 6.00 1.24 0.94 >64 5.80 1.25 0.97 苊烯 0~31 25.87 1.02 0.94 32~64 20.46 1.02 0.97 >64 18.77 1.03 0.95 苊 0~31 27.07 1.00 0.96 32~64 22.27 1.01 0.94 >64 17.27 1.02 0.95 芴 0~31 16.73 1.04 0.94 32~64 14.41 1.05 0.97 >64 14.90 1.04 0.96 菲 0~31 22.38 1.07 0.95 32~64 21.07 1.07 0.94 >64 16.91 1.10 0.96 蒽 0~31 29.12 1.07 0.96 32~64 22.61 1.09 0.97 >64 23.94 1.06 0.96 荧蒽 0~31 26.29 1.06 0.94 32~64 21.04 1.10 0.95 >64 24.32 1.09 0.96 芘 0~31 20.26 1.15 0.95 32~64 19.47 1.15 0.95 >64 16.81 1.15 0.95 苯并[a]蒽 0~31 26.46 1.14 0.96 32~64 18.96 1.14 0.94 >64 14.45 1.17 0.94 䓛 0~31 27.28 1.10 0.95 32~64 21.23 1.08 0.97 >64 23.33 1.06 0.96 苯并[b]荧蒽 0~31 23.11 1.20 0.98 32~64 19.42 1.21 0.96 >64 21.74 1.19 0.94 苯并[k]荧蒽 0~31 24.78 1.22 0.97 32~64 21.27 1.23 0.98 >64 18.23 1.24 0.98 苯并[a]芘 0~31 23.69 1.09 0.95 32~64 16.57 1.08 0.96 >64 14.63 1.11 0.96 二苯并[a,h]蒽 0~31 38.33 1.08 0.96 32~64 33.05 1.10 0.95 >64 28.15 1.09 0.95 茚并[1,2,3-cd]芘 0~31 18.01 1.34 0.94 32~64 15.68 1.38 0.96 >64 16.12 1.37 0.96 苯并[g,h,i]苝 0~31 10.25 1.20 0.96 32~64 9.50 1.20 0.95 >64 9.52 1.18 0.96 此外,在只考虑粒径因素的情况下,沉积物对萘的吸附能力最小,对二苯并[a, h]蒽的吸附能力最大(表4),其可能的原因是萘的挥发性较高,水体中的萘大多数挥发而难以被沉积物捕获所致。同时,0~31 μm的细颗粒沉积物的吸附能力K普遍高于32~64 μm的中粒径颗粒和>64 μm的粗粒径颗粒(图4)。3种粒径沉积物对同种PAHs的吸附能力相近,部分中粒径(32~64 μm)颗粒对于PAHs组分的吸附能力低于粗粒径(>64 μm)颗粒,如芴、蒽、荧蒽、䓛、苯并[b]荧蒽、苯并[a]芘、茚并[1, 2, 3-cd]芘和苯并[g, h, i]苝等,其余部分则是中粒径(32~64 μm)颗粒吸附能力高于粗粒径(>64 μm)颗粒(表4)。将16种PAHs按照环数分为低环、中环、高环3类,并通过室内等温吸附试验研究发现,不同的粒径颗粒沉积物对于PAHs的吸附能力有所差异,同等粒径颗粒沉积物对于不同环数PAHs的吸附能力也有较大差异(图5)。3种粒径颗粒对不同环数PAHs吸附能力为高环>低环>中环。一般而言,高环数PAHs的正辛醇-水分配系数相对较高,疏水性更强,因而更易被沉积物吸附。中环PAHs的累积吸附能力K在3种环数PAHs中最低,这是由于16种标准PAHs溶液中,低环(2+3环)与高环(5+6环)PAHs各有6种,中环(4环)PAHs则只有4种造成的。此外,相同环数PAHs的残留特征也存在差别,如高环(5+6环)PAHs 中,二苯并[a, h]蒽的吸附能力K明显高于其他高环数PAHs(表4),这可能与二苯并[a,h]蒽本身的物理化学性质有关。
前人研究认为沉积物吸附PAHs的主要影响因素有悬浮颗粒物、有机碳、pH、水动力学条件等[6-7]。本研究中,随着粒径颗粒增大,其对PAHs吸附能力也相应减弱(表3、图4),且TOC与PAHs含量间没有明显相关关系,样品采集于同一地点,故盐度和pH值被假设为定值,不会对颗粒吸附PAHs的行为产生差异化影响。因此,三种不同粒径对于PAHs的吸附能力不同主要为粒径因素的影响。粒径因素又包含颗粒的比表面积与孔隙直径。一般而言,不同粒径沉积物团聚的空隙结构存在显著差异,一般粗粒径颗粒通过微团聚体黏合而成,其中微团聚体呈单粒排列,因此粗粒径沉积物中的孔隙直径比细粒径沉积物更大[11]。本研究中,细粒径颗粒对于PAHs有着最大的吸附能力(图4),表明比表面积对PAHs的吸附起着主导作用。更大的比表面积,提供了更多的吸附点位,使得PAHs可以相对更多地暴露于沉积物表面,使得水体中的PAHs被大量吸附。相比而言,孔隙直径因素对PAHs的吸附起次要作用。更大的孔隙直径,使得PAHs分子更容易进入沉积物内部并被沉积物内部的吸附点位所吸附,因此部分粗粒径的沉积物颗粒对PAHs的吸附能力超过了中粒径颗粒(表3)。
总而言之,单一粒径因素对于PAHs的吸附作用存在影响,且粒径对于PAHs的吸附能力影响主要是由沉积物颗粒的比表面积所决定的,孔隙直径因素起次要作用。
3.4 Freundlich吸附实验反演
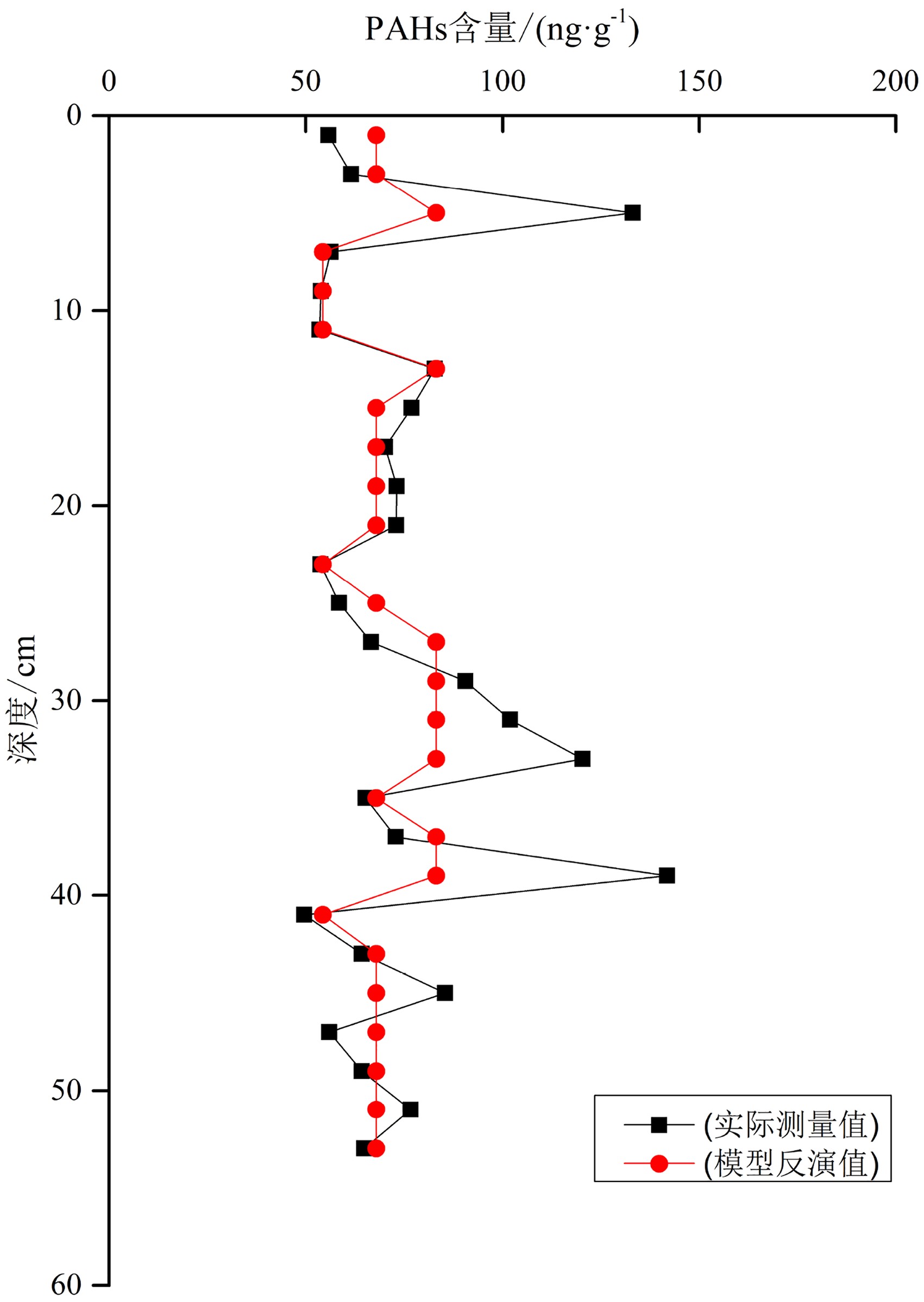
上文的模拟实验得到Freundlich等温式的参数n、K,通过达到平衡时水体中PAHs的浓度,就可以用来反演达到平衡时单位沉积物内PAHs的吸附量。然而由于研究区黄海近海表层水体中PAHs含量未见报道,因此本文采用长江河口表层水体平均的PAHs浓度值进行反演[29],所得结果见图6。反演结果和实际测量结果在大部分深度有着较强的一致性,而在某些深度差异明显,尤其是在5 cm深度以及30~40 cm深度处。这可能是在30~40 cm深度处样品的TOC含量相对较高(图2),TOC的芳香性组分可能对PAHs产生相似相容原理,吸附了更多的PAHs[30];而在上部5 cm则可能是由于生物的扰动作用,致使实际结果相对反演结果偏高。
除以上3处以外,总体而言,Freundlich等温模型模拟结果和实测结果有着较好的一致性。说明在沉积物有机质含量较低时,粒径是其控制吸附PAHs的主要影响因素。
4. 结论
(1)盐城潮滩沉积物中PAHs的含量为49.67~141.90 ng·g−1,平均为74.92 ng·g−1。研究区潮滩沉积物中PAHs污染处于低—中水平。
(2)不同环数 PAHs 对总浓度的贡献从大到小分别为:3环(24.55%)>4环(23.06%)>5环(20.34%)>2环(16.62%)>6环(15.43%),样品中以3—5环PAHs对总浓度的贡献率较高,表明研究区域 PAHs主要来自高温燃烧源。运用分子比值法及主成分分析法确定了研究区PAHs主要来源为各类燃烧源的混合来源。
(3)在只考虑粒径因素的情况下,盐城沉积物对萘的吸附能力最小,对二苯并[a, h]蒽的吸附能力最大。同时,在沉积物中有机质含量很低时,粒径为控制沉积物吸附PAHs的主要因素,并且粒径对于PAHs的吸附能力影响主要是由沉积物颗粒的比表面积所决定。
致谢:孙远见参与了野外调查与采样工作,评审老师为文章修改提出宝贵意见,于志腾(自然资源部第二海洋研究所)为文章修改提供帮助,谨致谢忱。
-
特征分子比 石油燃烧 煤炭燃烧 草木燃烧 石油源 荧蒽/(荧蒽+芘) 0.4~0.5 >0.5 >0.5 <0.4 苯并[a]蒽/(苯并[a]蒽+䓛) >0.35 >0.35 >0.35 <0.2 茚并芘/(茚并芘+苯并苝) 0.2~0.5 >0.5 >0.5 <0.2 表 2 沉积物中PAHs分子比值
Table 2 Isomeric ratios of PAHs in sediments
特征分子比 范围 平均值 指示意义 荧蒽/(荧蒽+芘) 0.44~0.55 0.51 燃烧源 苯并[a]蒽/(苯并[a]蒽+䓛) 0.27~0.49 0.41 燃烧源 茚并芘/(茚并芘+苯并苝) 0.41~0.53 0.49 石油燃烧源 表 3 PAHs组分在主成分上的因子载荷和累积方差
Table 3 The components load of PAHs (PC1) in sediment and the variance of PCA (PC2)
PAHs组分 PC1 PC2 萘 0.484 0.743 苊烯 0.798 0.435 苊 0.902 0.375 芴 0.905 0.105 菲 0.946 −0.053 蒽 0.955 −0.053 荧蒽 0.959 −0.227 芘 0.906 −0.301 苯并[a]蒽 0.986 −0.152 䓛 0.953 −0.278 苯并[b]荧蒽 0.957 −0.219 苯并[k]荧蒽 0.924 0.189 苯并[a]芘 0.983 −0.079 二苯并[a,h]蒽 0.946 0.104 茚并[1,2,3-cd]芘 0.969 −0.005 苯并[g,h,i]苝 0.967 −0.128 累积方差% 84.696 92.468 表 4 Freundlich模型下盐城潮滩三种粒径物质颗粒吸附能力参数
Table 4 Freundlich sorption parameters of PAHs with different sediment sizes from the Yancheng tidal flat
PAHs组分 粒径/μm K n R2 萘 0~31 8.09 1.22 0.96 32~64 6.00 1.24 0.94 >64 5.80 1.25 0.97 苊烯 0~31 25.87 1.02 0.94 32~64 20.46 1.02 0.97 >64 18.77 1.03 0.95 苊 0~31 27.07 1.00 0.96 32~64 22.27 1.01 0.94 >64 17.27 1.02 0.95 芴 0~31 16.73 1.04 0.94 32~64 14.41 1.05 0.97 >64 14.90 1.04 0.96 菲 0~31 22.38 1.07 0.95 32~64 21.07 1.07 0.94 >64 16.91 1.10 0.96 蒽 0~31 29.12 1.07 0.96 32~64 22.61 1.09 0.97 >64 23.94 1.06 0.96 荧蒽 0~31 26.29 1.06 0.94 32~64 21.04 1.10 0.95 >64 24.32 1.09 0.96 芘 0~31 20.26 1.15 0.95 32~64 19.47 1.15 0.95 >64 16.81 1.15 0.95 苯并[a]蒽 0~31 26.46 1.14 0.96 32~64 18.96 1.14 0.94 >64 14.45 1.17 0.94 䓛 0~31 27.28 1.10 0.95 32~64 21.23 1.08 0.97 >64 23.33 1.06 0.96 苯并[b]荧蒽 0~31 23.11 1.20 0.98 32~64 19.42 1.21 0.96 >64 21.74 1.19 0.94 苯并[k]荧蒽 0~31 24.78 1.22 0.97 32~64 21.27 1.23 0.98 >64 18.23 1.24 0.98 苯并[a]芘 0~31 23.69 1.09 0.95 32~64 16.57 1.08 0.96 >64 14.63 1.11 0.96 二苯并[a,h]蒽 0~31 38.33 1.08 0.96 32~64 33.05 1.10 0.95 >64 28.15 1.09 0.95 茚并[1,2,3-cd]芘 0~31 18.01 1.34 0.94 32~64 15.68 1.38 0.96 >64 16.12 1.37 0.96 苯并[g,h,i]苝 0~31 10.25 1.20 0.96 32~64 9.50 1.20 0.95 >64 9.52 1.18 0.96 -
[1] Yunker M B, Macdonald R W, Vingarzan R, et al. PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition [J]. Organic Geochemistry, 2002, 33(4): 489-515. doi: 10.1016/S0146-6380(02)00002-5
[2] Zhang Y X, Tao S. Global atmospheric emission inventory of polycyclic aromatic hydrocarbons (PAHs) for 2004 [J]. Atmospheric Environment, 2009, 43(4): 812-819. doi: 10.1016/j.atmosenv.2008.10.050
[3] Kim G B, Maruya K A, Lee R F, et al. Distribution and sources of polycyclic aromatic hydrocarbons in sediments from Kyeonggi Bay, Korea [J]. Marine Pollution Bulletin, 1999, 38(1): 7-15. doi: 10.1016/S0025-326X(99)80006-X
[4] Lin C, Liu J L, Wang R M, et al. Polycyclic aromatic hydrocarbons in surface soils of Kunming, China: concentrations, distribution, sources, and potential risk [J]. Soil and Sediment Contamination: An International Journal, 2013, 22(7): 753-766. doi: 10.1080/15320383.2013.768201
[5] Gao S, Collins M B. Holocene sedimentary systems on continental shelves [J]. Marine Geology, 2014, 352: 268-294. doi: 10.1016/j.margeo.2014.03.021
[6] 毕丽姣, 周岩梅, 张林林, 等. 太子河中游河段有机污染物分布特征研究[J]. 环境科学与技术, 2016, 39(S1):164-168 BI Lijiao, ZHOU Yanmei, ZHANG Linlin, et al. Distribution characteristics of organic contaminant in middle reaches of Taizihe river [J]. Environmental Science & Technology, 2016, 39(S1): 164-168.
[7] 王喆, 卢丽. 南宁市清水泉地下河水中多环芳烃分布特征[J]. 环境科学与技术, 2016, 39(4):132-136 WANG Zhe, LU Li. Distribution characteristics of polycyclic aromatic hydrocarbons in water from Qingshuiquan underground river in Nanning [J]. Environmental Science & Technology, 2016, 39(4): 132-136.
[8] Müller S, Wilcke W, Kanchanakool N, et al. Polycyclic aromatic hydrocarbons (PAHs) and polychlorinated biphenyls (PCBs) in particle-size separates of urban soils in Bangkok, Thailand [J]. Soil Science, 2000, 165(5): 412-419. doi: 10.1097/00010694-200005000-00005
[9] Wang B, Wang C, Huang Q Y, et al. Adsorption of PAHs on the sediments from the Yellow River delta as a function of particle size and salinity [J]. Soil and Sediment Contamination: An International Journal, 2015, 24(2): 103-115. doi: 10.1080/15320383.2014.920292
[10] Wang X C, Zhang Y X, Chen R F. Distribution and partitioning of polycyclic aromatic hydrocarbons (PAHs) in different size fractions in sediments from Boston Harbor, United States [J]. Marine Pollution Bulletin, 2001, 42(11): 1139-1149. doi: 10.1016/S0025-326X(01)00129-1
[11] 李锐龙, 刘贝贝, 朱亚先, 等. 芘在不同粒径红树林沉积物团聚体上的吸附/解吸行为[J]. 环境化学, 2014, 33(3):423-431 doi: 10.7524/j.issn.0254-6108.2014.03.012 LI Ruilong, LIU Beibei, ZHU Yaxian, et al. Sorption/desorption behaviors of pyrene on mangrove sediment aggregates with different sizes [J]. Environmental Chemistry, 2014, 33(3): 423-431. doi: 10.7524/j.issn.0254-6108.2014.03.012
[12] 李杨帆, 朱晓东, 邹欣庆, 等. 江苏盐城海岸湿地景观生态系统研究[J]. 海洋通报, 2005, 24 (4):46-51 doi: 10.3969/j.issn.1001-6392.2005.04.008 LI Yangfan, ZHU Xiaodong, ZOU Xinqing, et al. Study on landscape ecosystem of coastal wetlands in Yancheng, Jiangsu Province [J]. Marine Science Bulletin, 2005, 24 (4): 46-51. doi: 10.3969/j.issn.1001-6392.2005.04.008
[13] Du J B, Shi B W, Li J S, et al. Muddy coast off Jiangsu, China: physical, ecological, and anthropogenic processes[M]//. Wang X H. Sediment Dynamics of Chinese Muddy Coasts and Estuaries. Boston: Academic Press, 2019.
[14] 成都地质学院陕北队. 沉积岩(物)粒度分析及其应用[M]. 北京: 地质出版社, 1978. North Shaanxi Team of Chengdu University of Geology. Analysis and application of grain size[M]. Beijing: Geological Publishing House, 1978.
[15] 鲁如坤. 土壤农业化学分析方法[M]. 北京: 中国农业科技出版社, 2000. LU Rukun. Methods of soil agricultural chemistry analyses[M]. Beijing: China Agricultural Science and Technology Press, 2000.
[16] 中华人民共和国环境保护局. HJ 805-2016, 土壤和沉积物多环芳烃的测定 气相色谱-质谱法[S]. 北京: 中国环境科学出版社, 2016. Environmental Protection Bureau of the People's Republic of China. HJ 805-2016, Soil and sediment–determination of polycyclic aromatic hydrocarbon by gas chromatography-mass spectrometry method[S]. Beijing: China Environmental Science Press, 2016.
[17] 中华人民共和国国家质量监督检验检疫总局, 中国国家标准化管理委员会. GB/T 26411-2010, 海水中16种多环芳烃的测定 气相色谱-质谱法[S]. 北京: 中国标准出版社, 2011. General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China GB/T 26411-2010, Determination of 16 polynuclear aromatic hydrocarbons in seawater by GC-MS[S]. Beijing: Standards Press of China, 2011.
[18] Wang Y, Shen C C, Shen Z Y, et al. Spatial variation and sources of polycyclic aromatic hydrocarbons (PAHs) in surface sediments from the Yangtze Estuary, China [J]. Environmental Science: Processes & Impacts, 2015, 17(7): 1340-1347.
[19] Mead, J. A. A comparison of the Langmuir, Freundlich and Temkin equations to describe phosphate adsorption properties of soils [J]. Australian Journal of Soil Research, 1981, 19(3): 333-342. doi: 10.1071/SR9810333
[20] Baumard P, Budzinski H, Garrigues P. Polycyclic aromatic hydrocarbons in sediments and mussels of the western Mediterranean Sea [J]. Environmental Toxicology and Chemistry, 1998, 17(5): 765-776. doi: 10.1002/etc.5620170501
[21] Bjørseth A, Ramdahl T. Handbook of Polycyclic Aromatic Hydrocarbons, Volume 2, Emission Sources and Recent Progress in Analytical Chemistry[M]. New York: Marcel Dekker, 1985.
[22] 郭俊丽, 杨旸, 高建华, 等. 浙闽沿岸泥质区南部表层沉积物中多环芳烃的分布、来源及生态风险评价[J]. 海洋环境科学, 2017, 36(6):826-831 GUO Junli, YANG Yang, GAO Jianhua, et al. The distribution, sources and ecological risk assessment of surficial sediment in the south coastal mud Area off Zhejiang-Fujian [J]. Marine Environmental Science, 2017, 36(6): 826-831.
[23] Wilcke W. Synopsis polycyclic aromatic hydrocarbons (PAHs) in soil—a review [J]. Journal of Plant Nutrition and Soil Science, 2000, 163(3): 229-248. doi: 10.1002/1522-2624(200006)163:3<229::AID-JPLN229>3.0.CO;2-6
[24] Fernandes M B, Sicre M A, Boireau A, et al. Polyaromatic hydrocarbon (PAH) distributions in the Seine River and its estuary [J]. Marine Pollution Bulletin, 1997, 34(11): 857-867. doi: 10.1016/S0025-326X(97)00063-5
[25] Yan B Z, Abrajano T A, Bopp R F, et al. Molecular tracers of saturated and polycyclic aromatic hydrocarbon inputs into Central Park Lake, New York City [J]. Environmental Science & Technology, 2005, 39(18): 7012-7019.
[26] Sofowote U M, McCarry B E, Marvin C H. Source apportionment of PAH in Hamilton Harbour suspended sediments: comparison of two factor analysis methods [J]. Environmental Science & Technology, 2008, 42(16): 6007-6014.
[27] Wang C H, Wu S H, Zhou S L, et al. Polycyclic aromatic hydrocarbons in soils from urban to rural areas in Nanjing: Concentration, source, spatial distribution, and potential human health risk [J]. Science of the Total Environment, 2015, 527-528: 375-383. doi: 10.1016/j.scitotenv.2015.05.025
[28] Simoneit B R T. Biomass burning—a review of organic tracers for smoke from incomplete combustion [J]. Applied Geochemistry, 2002, 17(3): 129-162. doi: 10.1016/S0883-2927(01)00061-0
[29] 范博, 王晓南, 黄云, 等. 我国七大流域水体多环芳烃的分布特征及风险评价[J]. 环境科学, 2019, 40(5):2101-2114 FAN Bo, WANG Xiaonan, HUANG Yun, et al. Distribution and risk assessment of polycyclic aromatic hydrocarbons in water bodies in seven basins of China [J]. Environmental Science, 2019, 40(5): 2101-2114.
[30] 丁爱芳. 江苏省部分地区农田土壤中多环芳烃(PAHs)的分布与生态风险[D]. 南京农业大学博士学位论文, 2007. DING Aifang. Distribution of polycyclic aromatic hydrocarbons (PAHs) in farm-land of parts of Jiangsu Province and its ecological risk[D]. Doctor Dissertation of Nanjing Agricultural University, 2007.
-
期刊类型引用(2)
1. 闫施帅,鄢全树,石学法,袁龙. 海底高原与俯冲带相互作用的地质效应的研究进展. 地球科学进展. 2024(07): 702-716 .  百度学术
百度学术
2. 鄢全树,袁龙,闫施帅,刘振轩,吴增,石学法. 菲律宾海板块东南边界地质过程与研究展望. 海洋地质与第四纪地质. 2023(05): 50-63 .  本站查看
本站查看
其他类型引用(0)



 下载:
下载: